- 蛋白纯度检测
实体瘤靶点蛋白
近年来,随着生物制药市场的快速增长,与小分子药物相比,抗体药物在特异性和选择性方面表现更高。因此,在血液肿瘤和自身免疫性疾病的治疗方面取得了显著进展。然而,由于实体瘤复杂的免疫微环境,针对实体瘤的治疗性抗体药物开发仍然面临更大的挑战。

实体瘤占全球癌症发病率的大多数,仍然是抗体药物、抗体偶联药物(ADC)及细胞治疗开发的主要方向。靶点发现的研究越来越集中于肿瘤特异性表面抗原、肿瘤微环境相关蛋白和驱动致癌因素。功能性抗体必须在体内发挥活性,因此识别具有功能性的单克隆抗体至关重要。
缔码生物在免疫原蛋白开发过程中非常重视质量控制。所有蛋白均采用 HEK293 哺乳动物细胞分泌表达系统制备,并实施严格的质量控制,包括纯度检测、抗体‑药物相互作用验证、冻融稳定性测试、热稳定性测试等。
重点实体瘤靶点
肿瘤相关膜蛋白是单克隆抗体、抗体偶联药物(ADC)、双特异性抗体以及 CAR‑T 细胞疗法的主要靶点。以下列出一些具有代表性且备受关注的实体瘤靶点:
Claudin 18.2 (CLDN18.2)
特异性表达于胃癌和胰腺癌中的紧密连接蛋白,是ADC和CAR‑T研发中的重要靶点。
Mesothelin (MSLN)
在间皮瘤、卵巢癌和胰腺癌中高表达,常作为抗体和细胞治疗的靶标。
应用场景
- 用于抗体药物开发的免疫原
- 体外或体内功能实验试剂
- 药物先导分子验证试剂
- 抗体亲和力检测实验
质控数据
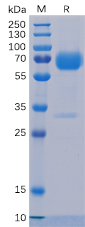
Figure 1. Human Trop2 Protein, mFc-His Tag (PME100501) on SDS-PAGE under reducing condition.
- 蛋白活性检测
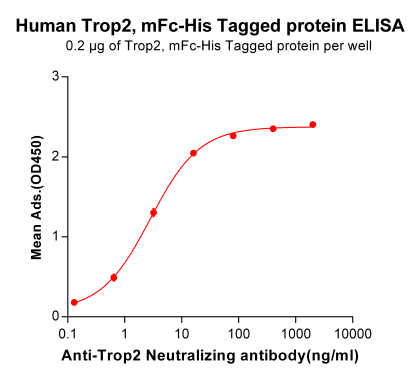
Figure 2. Immoblized Human Trop2 protein (PME100501) can bind Anti-Trop2 Neutralizing antibody (BME100023) in a linear range of 0.13-16.0 ng/ml.
热门实体瘤靶点列表
| ALK | AXL | CEACAM5 | CLDN18.2 | CLDN6 | EGFR | EPCAM | FAP |
| FGFR2 | FGFR3 | HER2 | KRAS | MET | MSLN | MUC1 | NECTIN4 |
| NTRK1 | PSCA | PTK7 | RET | ROR1 | ROR2 | TGFBR2 | TROP2 |
| VEGFA | YAP1 |


